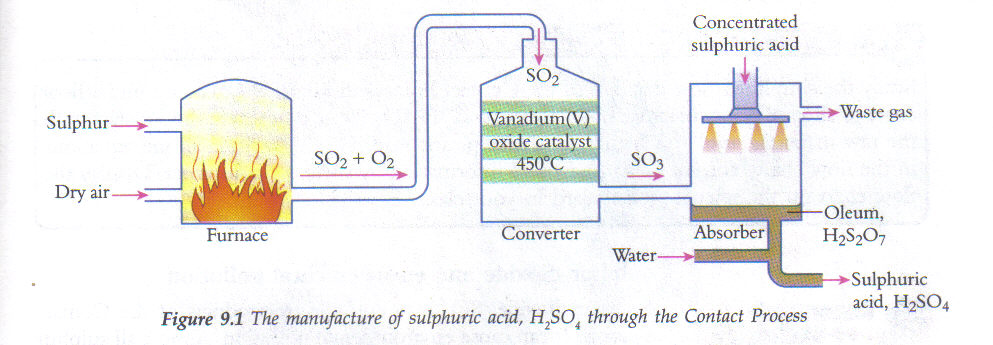
The contact process in the manufacture of sulfuric acid involves five primary steps:
2) Purification of sulfur dioxide to remove dust particles, oxides of iron and arsenic. The purification unit includes a series of stages:
Dusting Tower:
The mixture of sulfur dioxide and excess of air contains several impurities that must be removed as it may result in loss of efficiency of the catalyst. The sulfur dioxide is mixed with an excess of air and is passed through a purifier in which electric charges attract the solid particles that are removed.
Washing Tower:
In this stage, sulfur dioxide is washed completely to rinse away any soluble contaminants
Drying Tower:
In the stage, the gas is dried by a spray of concentrated sulfuric acid for further purification
Arsenic Purifier:
In this stage, the gaseous mixture is directed into the arsenic purifier to remove arsenic oxide
Once the sulfuric dioxide is completely purified, it can move onto the next step which is the conversion to sulfuric trioxide.
Dusting Tower:
The mixture of sulfur dioxide and excess of air contains several impurities that must be removed as it may result in loss of efficiency of the catalyst. The sulfur dioxide is mixed with an excess of air and is passed through a purifier in which electric charges attract the solid particles that are removed.
Washing Tower:
In this stage, sulfur dioxide is washed completely to rinse away any soluble contaminants
Drying Tower:
In the stage, the gas is dried by a spray of concentrated sulfuric acid for further purification
Arsenic Purifier:
In this stage, the gaseous mixture is directed into the arsenic purifier to remove arsenic oxide
Once the sulfuric dioxide is completely purified, it can move onto the next step which is the conversion to sulfuric trioxide.
3) Converting sulphur dioxide into sulphur trioxide (Applying Le Chatelier's principle)
In order to manufacture industrial sulphuric acid, the contact process of sulfur trioxide production has to be economically efficient.
In order to manufacture industrial sulphuric acid, the contact process of sulfur trioxide production has to be economically efficient.
To convert sulphur dioxide to sulphur trioxide, sulphur dioxide is mixed with oxygen and the mixture is passed over a catalyst of Vanadium (V) oxide (V2O5) at a temperature as high as 450C and at a pressure of 2 atm. Mixture of sulphur dioxide and oxygen forms sulphur trioxide. At standard conditions, the equilibrium is positioned to the left and the amount of sulphur trioxide (SO3) produced is relatively small. In order to improve the yield of sulfur trioxide, the reaction is carried out at 450C and 2 atmospheric pressure using catalysts compounds such as Vanadium oxide (V2O5).
2SO2(g) + O2(g) <---> 2SO3(g) (ΔH = -196 kJ/mol-1)
Kc = [SO3]2
[SO2]2 [O2]1
This is the primary chemical reaction that occurs in the Contact process in which it is an reversible, an exothermic reaction.
Temperature:
If the temperature is increased, the rate of reaction increases due to more collision per unit time and high energy collisions, and thus the equilibrium will shift to the endothermic side (reactants) resulting in less sulfur trioxide at a fast reaction rate. If the temperature is decreased, the rate of reaction will be slower but it will produce more sulfur trioxide since the equilibrium will shift to the exothermic side (products). Hence why the reaction is carried out at a more optimal temperature (450C)
Pressure:
Since only two atmospheric pressure is used, the pressure is relatively low, therefore it favours the reactants side with the most gas particles in order to increase the pressure since it is a low pressure reaction. A pressure of 2 atm is sufficient enough to obtain a great yield, so it isn't necessary to spend money on expensive resistant vessels and resources to increase the pressure.
Catalysts:
Vanadium oxide (V2O5) is used as catalyst in this reaction which increases the rate of reaction and reduces the need for a high temperature.
4) Conversion of SO3 to oleum
SO3(g) + H2SO4(l) --> H2S2O7(l) - oleum
In this step of the contact process, concentrated sulfuric acid is used to dissolve the sulfur trioxide which produces the oleum. Sulphur trioxide is cooled in a heat exchanger and is then absorbed in concentrated sulphuric acid in another tower to provide oleum with H2S2O7.
5) Dilution of oleum to sulfuric acid
H2S2O7 + H2O --> 2H2SO4
In this final step of the contact process, dilution of oleum is done by reacting it with water, and as a result, concentrated sulphuric acid is produced (98% H2SO4).The reaction to form sulphuric acid solution is exothermic with a large K value, therefore if the sulphur trioxide is added to water, an acid mist forms and is difficult to contain.
2SO2(g) + O2(g) <---> 2SO3(g) (ΔH = -196 kJ/mol-1)
Kc = [SO3]2
[SO2]2 [O2]1
This is the primary chemical reaction that occurs in the Contact process in which it is an reversible, an exothermic reaction.
Temperature:
If the temperature is increased, the rate of reaction increases due to more collision per unit time and high energy collisions, and thus the equilibrium will shift to the endothermic side (reactants) resulting in less sulfur trioxide at a fast reaction rate. If the temperature is decreased, the rate of reaction will be slower but it will produce more sulfur trioxide since the equilibrium will shift to the exothermic side (products). Hence why the reaction is carried out at a more optimal temperature (450C)
Pressure:
Since only two atmospheric pressure is used, the pressure is relatively low, therefore it favours the reactants side with the most gas particles in order to increase the pressure since it is a low pressure reaction. A pressure of 2 atm is sufficient enough to obtain a great yield, so it isn't necessary to spend money on expensive resistant vessels and resources to increase the pressure.
Catalysts:
Vanadium oxide (V2O5) is used as catalyst in this reaction which increases the rate of reaction and reduces the need for a high temperature.
4) Conversion of SO3 to oleum
SO3(g) + H2SO4(l) --> H2S2O7(l) - oleum
In this step of the contact process, concentrated sulfuric acid is used to dissolve the sulfur trioxide which produces the oleum. Sulphur trioxide is cooled in a heat exchanger and is then absorbed in concentrated sulphuric acid in another tower to provide oleum with H2S2O7.
5) Dilution of oleum to sulfuric acid
H2S2O7 + H2O --> 2H2SO4
In this final step of the contact process, dilution of oleum is done by reacting it with water, and as a result, concentrated sulphuric acid is produced (98% H2SO4).The reaction to form sulphuric acid solution is exothermic with a large K value, therefore if the sulphur trioxide is added to water, an acid mist forms and is difficult to contain.