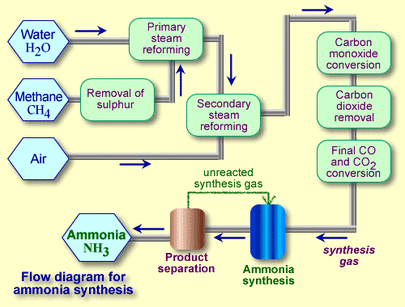
The main industrial process to produce ammonia with the reaction of nitrogen gas and hydrogen gas. It is an important process because ammonia is difficult to produce and the fertilizer created from the ammonia is responsible for sustaining one-third of the Earth's population.
Ammonia is produced in the chemical process by reacting nitrogen gas with hydrogen gas. The formation of ammonia is a reversible and exothermic reaction as heat is released to its surroundings, giving off 46kJ of heat per mole of ammonia formed.
N2(g) + 3 H2(g) → 2 NH3(g) (ΔH = -92 kJ mol-1)
The equilibrium law expression is:
Kc = [NH3]2
[N2]1 [H2]3
The equilibrium law expression is:
Kc = [NH3]2
[N2]1 [H2]3
Applying Le Chatelier's Principle in the Production of Ammonia
At normal conditions, the equilibrium is positioned to the left and the amount of ammonia formed is quite insufficient, therefore, to significantly increase the yield of ethanol, the reaction is carried out at temperatures between 400-450C C and 200 atmospheric pressure using catalyst compounds such as iron oxide (Fe3O4) which acts as a catalyst in order to speed up the reaction efficiently.
Temperature:
The forward reaction is exothermic which means that the the position of the equilibrium must be shifted to the right in order to produce the maximum amount of ammonia. According to Le Chatelier's Principle, by lowering the temperature, the system will move the position of the equilibrium to the right to initially counteract this by producing more heat, allowing for more ammonia to be produced.
Compromise:
Although the economic compromise is at a higher temperature at 400-450C, the rate of reaction increases significantly and the equilibrium shifts to the reactants side, resulting in less yield of ammonia.It is sufficient enough to produce high proportions of ammonia in a very short time. This is better than attempting to achieve an equilibrium mixture obtaining a high yield of ammonia at an extremely slow reaction rate, hence why the reaction is carried out at 450C.
Pressure:
There are four gas molecules on the reactants side and only two gas molecules on the right side (4>2), which means there is a net decrease in gas molecules in producing ammonia. According to Le Chatelier's principle, increasing the pressure would shift the position of equilibrium to the side with fewer molecules to consequently reduce the pressure since the system wants to bring the pressure back down by decreasing the molecules. To obtain the maximum yield of ammonia, high pressure is required as it increases higher collisions of molecules and in this case, 200 atmospheres is relatively high.
Disadvantages of high pressure:
It is expensive since extremely strong pipes and resistant vessels are necessary to withstand high pressures and is also more dangerous to address the health and safety issues involved.
Catalysts:
Lower temperatures results in the process being slow which is why catalysts such as Iron Oxide (Fe3O4)is used to initially speed the rate of reaction but does not effect the position of equilibrium at all, which is why manufacturers use catalysts.
Concentration:
In order to shift the equilibrium to the right, we can increase the concentration of the reactants or decrease the concentration of the products. According to Le Chatelier's Principle, when increasing the concentration on either the products or reactants, the equilibrium will shift in the opposite direction. So if we add more concentration to the reactants side, the equilibrium will shift towards the products, however if we decrease the concentration of ammonia, the position of equilibrium will move so that the concentration of ammonia increases again, this means that more reactants will react to replace the ammonia that has been removed, so the equilibrium will shift towards the right.
At normal conditions, the equilibrium is positioned to the left and the amount of ammonia formed is quite insufficient, therefore, to significantly increase the yield of ethanol, the reaction is carried out at temperatures between 400-450C C and 200 atmospheric pressure using catalyst compounds such as iron oxide (Fe3O4) which acts as a catalyst in order to speed up the reaction efficiently.
Temperature:
The forward reaction is exothermic which means that the the position of the equilibrium must be shifted to the right in order to produce the maximum amount of ammonia. According to Le Chatelier's Principle, by lowering the temperature, the system will move the position of the equilibrium to the right to initially counteract this by producing more heat, allowing for more ammonia to be produced.
Compromise:
Although the economic compromise is at a higher temperature at 400-450C, the rate of reaction increases significantly and the equilibrium shifts to the reactants side, resulting in less yield of ammonia.It is sufficient enough to produce high proportions of ammonia in a very short time. This is better than attempting to achieve an equilibrium mixture obtaining a high yield of ammonia at an extremely slow reaction rate, hence why the reaction is carried out at 450C.
Pressure:
There are four gas molecules on the reactants side and only two gas molecules on the right side (4>2), which means there is a net decrease in gas molecules in producing ammonia. According to Le Chatelier's principle, increasing the pressure would shift the position of equilibrium to the side with fewer molecules to consequently reduce the pressure since the system wants to bring the pressure back down by decreasing the molecules. To obtain the maximum yield of ammonia, high pressure is required as it increases higher collisions of molecules and in this case, 200 atmospheres is relatively high.
Disadvantages of high pressure:
It is expensive since extremely strong pipes and resistant vessels are necessary to withstand high pressures and is also more dangerous to address the health and safety issues involved.
Catalysts:
Lower temperatures results in the process being slow which is why catalysts such as Iron Oxide (Fe3O4)is used to initially speed the rate of reaction but does not effect the position of equilibrium at all, which is why manufacturers use catalysts.
Concentration:
In order to shift the equilibrium to the right, we can increase the concentration of the reactants or decrease the concentration of the products. According to Le Chatelier's Principle, when increasing the concentration on either the products or reactants, the equilibrium will shift in the opposite direction. So if we add more concentration to the reactants side, the equilibrium will shift towards the products, however if we decrease the concentration of ammonia, the position of equilibrium will move so that the concentration of ammonia increases again, this means that more reactants will react to replace the ammonia that has been removed, so the equilibrium will shift towards the right.
At the end of the process, when the gases are released from the iron catalyst, the gases is cooled under high pressure and ammonia liquefies as a liquid, whereas hydrogen and nitrogen remain as gases and is then recycled back through the reaction chamber.
Ammonia is commonly used in today's society and is essential for biological processes in which it plays a significant role in the nitrogen cycle. It is an important process because ammonia is used as a plant fertilizer, allowing farmers to grow a sufficient amount of crops to support society's increasing population. However, there are some negative impacts associated with the Haber process and that is when ammonia is exposed to humans, it can be very corrosive and harmful. It has many major purposes as most of it is used as a fertilizer, water purification, manufacturer of plastics and many more chemicals related, which is solely why equilibrium is manipulated to shift more to the right in order to increase the production of ammonia.
Ammonia is commonly used in today's society and is essential for biological processes in which it plays a significant role in the nitrogen cycle. It is an important process because ammonia is used as a plant fertilizer, allowing farmers to grow a sufficient amount of crops to support society's increasing population. However, there are some negative impacts associated with the Haber process and that is when ammonia is exposed to humans, it can be very corrosive and harmful. It has many major purposes as most of it is used as a fertilizer, water purification, manufacturer of plastics and many more chemicals related, which is solely why equilibrium is manipulated to shift more to the right in order to increase the production of ammonia.